“Ozone” was President George H.W. Bush’s pet name for Al Gore during the campaign in ’92. At that time, Gore’s weirdo environmental views were already well-known, but, that’s beside the point of this article.
What do we know about ozone?
 To better understand that question, we must discuss oxygen. Earth’s atmosphere is composed of 78% nitrogen, 21% oxygen and 1% “other gasses” which include water vapor, carbon dioxide and ozone. Included in the latter 1% is the dreaded “greenhouse gasses” you hear about. Less than one percent of the atmosphere is causing all that “trouble” — hmmmm. I digress — Back to oxygen . . .
To better understand that question, we must discuss oxygen. Earth’s atmosphere is composed of 78% nitrogen, 21% oxygen and 1% “other gasses” which include water vapor, carbon dioxide and ozone. Included in the latter 1% is the dreaded “greenhouse gasses” you hear about. Less than one percent of the atmosphere is causing all that “trouble” — hmmmm. I digress — Back to oxygen . . .
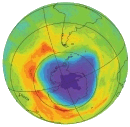
Right: The Antarctic ozone hole (Courtesy NASA/JPL-Caltech)
Because of the electrical structure of oxygen atoms, they tend to pair off in oxygen molecules — a molecule being two or more atoms combined — a diatomic molecule, O2, is the result. When electrical or radiation energy interacts with oxygen molecules, they split into atoms again and recombine as a triatomic molecule, O3, ozone. Ozone won’t last long in it’s triatomic state without constant application of an energy source.
Other facts about ozone:
- Ground level ozone is an air pollutant with harmful effects on lung function. It inhibits photosynthesis in plant-life and is corrosive to most metals. Ground-level ozone is caused by both natural and industrial activities.
- In the upper atmosphere, ozone prevents most (but not all) damaging ultraviolet light from reaching the Earth’s surface. Upper atmospheric ozone occurs almost entirely because of solar radiation.
- The “ozone hole” is a natural phenomenon that occurs near the magnetic south pole of Earth. The ozone hole grows with summer sunlight and shrinks during the dark Antarctic winter with its largest aperture occurring in September and smallest in March.
What about ozone-layer depletion?
Back in the 1980’s, it was discovered that chlorofluorocarbon gasses (CFC’s) would force O3 molecules to revert to O2 molecules, thus allowing the theory to develop that these gasses were depleting the ozone in the upper atmosphere. The ozone hole was the phenomenon most touted as an indication that this depletion was true. However, there has never been an absolute conclusion made to prove the connection.
CFCs are not the only things that can influence the ozone layer; sunspots, volcanoes and weather also play a role. Ultraviolet rays from sunspots boost the ozone layer, while sulfurous gases emitted by some volcanoes can weaken it.
Many people buy into these non-proven hypotheses as fact when the jury is still out on the matter. Science requires that hypotheses must dictate a series of tests which are then used to either prove or disprove the hypothesis. Any “consensus” science is merely faith — a belief in what the believer wants to be true.
Even NASA scientists still regard the ozone question as a puzzle:
Think of the ozone layer as Earth’s sunglasses, protecting life on the surface from the harmful glare of the sun’s strongest ultraviolet rays, which can cause skin cancer and other maladies.
People were understandably alarmed, then, in the 1980s when scientists noticed that manmade chemicals in the atmosphere were destroying this layer. Governments quickly enacted an international treaty, called the Montreal Protocol, to ban ozone-destroying gases such as CFCs then found in aerosol cans and air conditioners.
Today, almost 20 years later, reports continue of large ozone holes opening over Antarctica, allowing dangerous UV rays through to Earth’s surface. Indeed, the 2005 ozone hole was one of the biggest ever, spanning 24 million sq km in area, nearly the size of North America.
Listening to this news, you might suppose that little progress has been made. You’d be wrong.
